4 Questions Nanoparticle Researchers Are Really Asking About
2026-03-25News
4 Questions Nanoparticle Researchers Are Really Asking About
Even experienced nanoparticle researchers often circle back to the same core questions about how and why measurement results differ. Subtle changes in analytical technique, sample environment, and instrument configuration can transform the data, and ultimately, how we interpret the material itself. Drawing on Bettersize’s expertise in nanoparticle characterization, this article explores four recurring questions that underpin high-quality nanoscience, connecting principles of physical chemistry with the realities of day-to-day lab work.
1. Why Do SEM and DLS Give Different Particle Sizes?
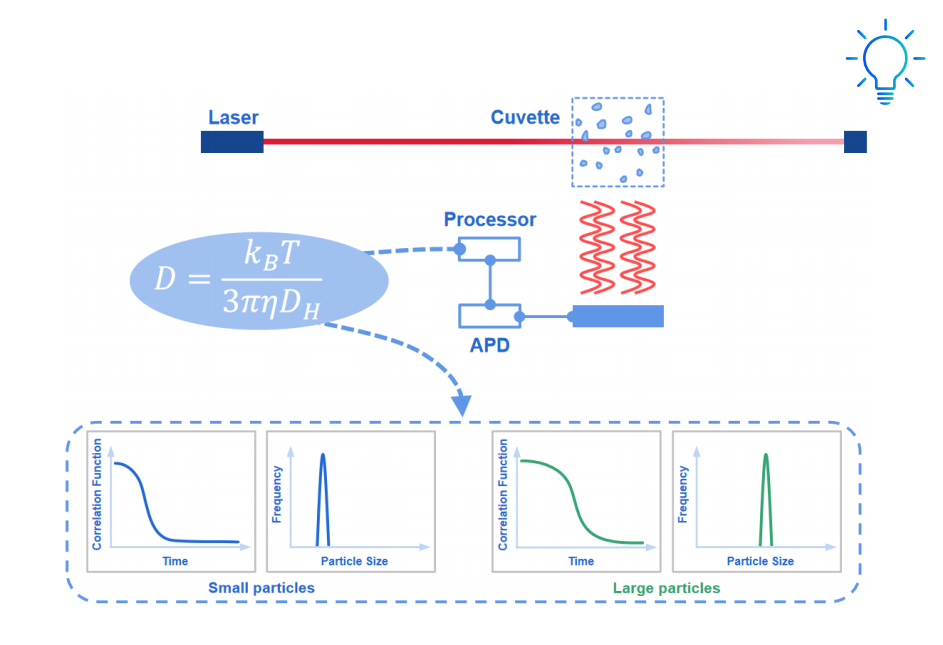
Scanning electron microscopy (SEM) and dynamic light scattering (DLS) are complementary, not contradictory. The key lies in what each technique measures. SEM reveals the projected diameter of individual particles under high vacuum, providing nanoscale resolution of shape and morphology. Because the sample is dried and often coated with a conductive layer, the particles lose their solvation shells, resulting in an apparent size smaller than that observed in suspension.
DLS, on the other hand, measures the hydrodynamic diameter; the effective radius of a particle diffusing through a liquid. This value encompasses the solid core, the surrounding solvent layer, and any adsorbed molecules such as surfactants or polymers. The result is often larger because DLS captures how the particle behaves dynamically, accounting for Brownian motion, the Stokes–Einstein relationship, and the electrical double layer that forms around charged surfaces. Swelling or soft coatings can further increase apparent size.
Brownian Motion
When viewed together, SEM and DLS offer a fuller picture: morphology and surface texture from SEM, and colloidal stability and dispersion behavior from DLS. Rather than conflicting, they describe different physical realities of the same system.
2. Is 173° Optics Superior to 90° Optics for Size Measurement?
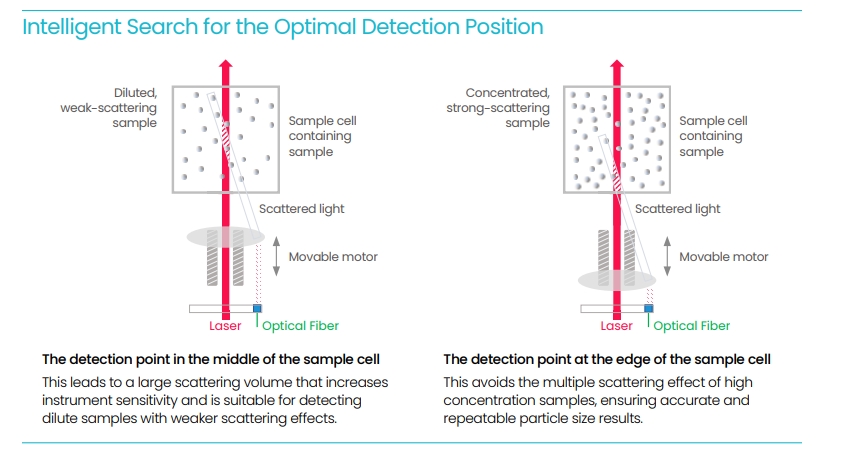
There is no single “best” scattering angle, only the most suitable one for a given sample. A 173° backscattering geometry offers superior sensitivity for weakly scattering nanoparticles. Because detection occurs close to the incident beam, it reduces multiple scattering and allows measurement at higher concentrations without excessive signal loss. This configuration also improves the signal-to-noise ratio, particularly for small particles and turbid dispersions.
By contrast, a 90° side-scattering setup requires a much smaller sample volume, sometimes as little as 3 μL, and is advantageous when sample availability is limited. While 173° optics excel in handling concentrated suspensions or proteins, 90° detection provides flexibility and convenience for routine checks and low-volume experiments.
In practice, researchers often use both geometries strategically. The ability to select between backscattering and side-scattering in a single instrument enables precision across a wide range of concentrations and particle sizes. This optical versatility is central to modern photon correlation spectroscopy and to ensuring reproducibility across labs.
3. Which Factors Affect the Zeta Potential of Nanoparticles?
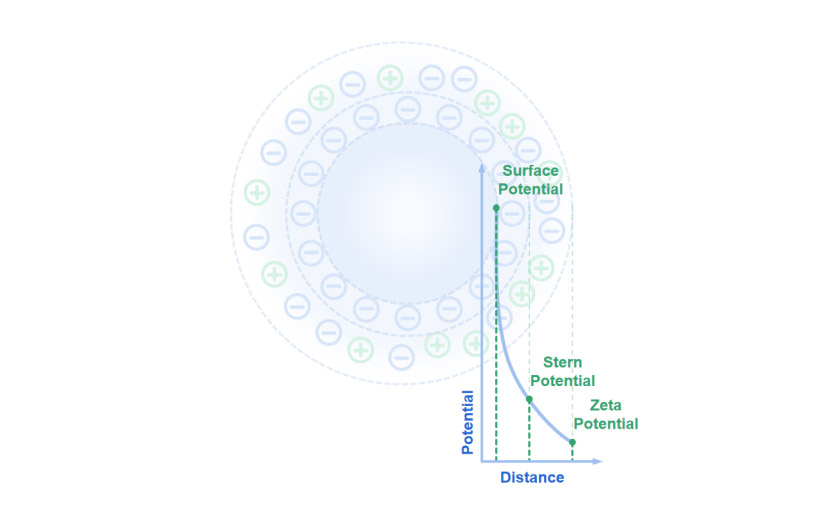
Zeta potential reflects the electrokinetic potential at the slipping plane of a colloidal particle, and it is highly sensitive to the surrounding chemical environment. The ionic strength, pH, surfactants, dispersant polarity, and particle concentration all affect the electric double layer that governs interparticle forces.
When ionic strength rises, due to salts or buffer ions, the double layer compresses, lowering the absolute value of the zeta potential. A pH shift alters surface charge through protonation or deprotonation of functional groups, moving the system toward or away from the isoelectric point. Surfactants and polymers, meanwhile, can adsorb to the surface and either neutralize or invert charge, introducing steric hindrance that modifies colloidal stability. Even the polarity and dielectric constant of the dispersant affect how counterions arrange themselves around each nanoparticle.
These effects are well described by DLVO theory, which balances electrostatic repulsion against van der Waals attraction. A high magnitude of zeta potential, positive or negative, suggests strong repulsion and thus a stable dispersion, whereas low values imply a tendency to aggregate. Controlling these parameters is key to engineering stable nanofluids, emulsions, and coatings.
4. What Are Thermal Stability Studies—and How Are They Performed?
Thermal stability testing examines how particle size, polydispersity index (PDI), and zeta potential evolve as temperature changes. By heating the dispersion in controlled increments and performing DLS and electrophoretic light scattering (ELS) at each step, researchers can observe whether nanoparticles maintain structural integrity or begin to aggregate, swell, or lose charge.
During such experiments, each temperature point is allowed to equilibrate before measurement to ensure thermal uniformity and consistent viscosity. Gradual increases in hydrodynamic size or sudden drops in zeta potential signal instability, often linked to phase transitions, polymer collapse, or loss of surfactant coverage.
This stepwise approach reveals critical information about formulation robustness. For nanomedicines, emulsions, and advanced composite materials, understanding how thermal stress influences colloidal behavior is essential for predicting storage performance, processing behavior, and long-term reliability.
From Measurement to Meaning
Every characterization technique captures a different aspect of nanoparticle behavior. SEM exposes the solid-state structure and surface topology. DLS reveals the hydrodynamic motion of solvated particles. Zeta potential analysis quantifies the invisible electrostatic forces that dictate colloidal stability. Thermal ramping experiments, in turn, uncover how these interactions evolve under stress.
When combined, these measurements provide more than numbers, they offer insight into how structure, surface chemistry, and environment work together to define nanoscale function. By integrating high-precision optics, automated temperature control, and advanced signal processing, Bettersize instruments enable nanoparticle researchers to move beyond isolated measurements and toward a coherent understanding of material dynamics, turning every data point into meaningful discovery.
Recent News